The first doses of the Pfizer-BioNTech COVID-19 vaccine were administered to hospital staff in Seattle Dec. 15.
The vaccine comes at a time when virus infections have reached record levels in Pierce County and some hospitals are at or approaching capacity, according to the state Department of Health.
“Our hospitals are stressed, taking care of patients, with or without COVID, and our health care workers are exhausted,” said Dr. Kathy Lofy, state health officer and the chief science officer for DOH.
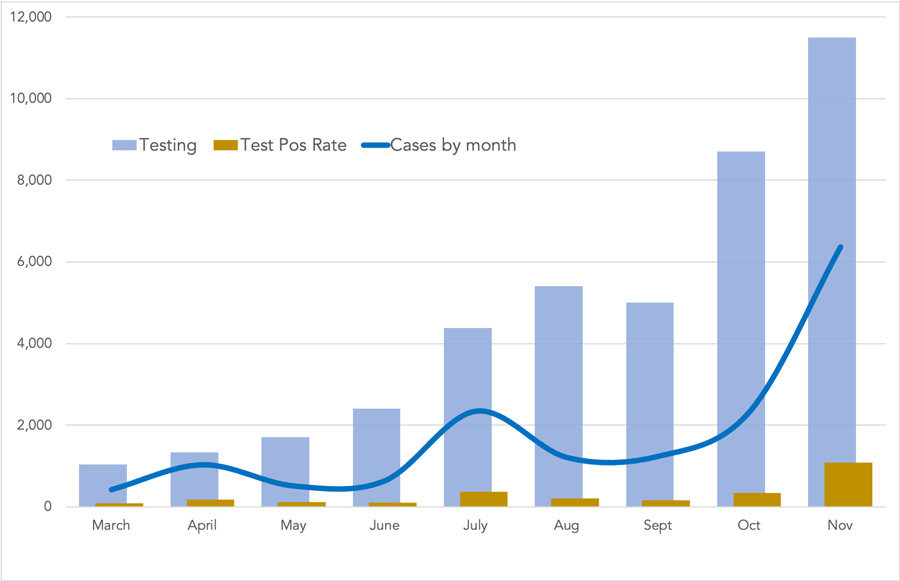
Hospitals in Pierce County were at an average 86% capacity by press time; anything over 80% is considered critical. St. Anthony in Gig Harbor was at 100% capacity, with 10% COVID-19 patients. Tacoma General was at 93% and 17%; St. Joseph Medical Center in Tacoma was at 60% and 9%.
Washington state expects to receive 192,750 doses of Pfizer vaccine by the end of December.
The Pfizer vaccine was approved by the FDA Dec. 11 and western states the next day for persons 16 years and older. Pediatric clinical trials continue, as do three other large-scale trials in the U.S. for additional COVID-19 vaccines.
A vaccine from the National Institutes of Health and Moderna was approved Dec. 18, which could mean another 183,000 doses available to Washington before year’s end. An additional 500,000 or more should arrive in January. The number of adults in each state determines the weekly delivery size.
The first people to receive vaccines will be health care workers and first responders directly involved with COVID-19 patients, long-term care facilities, and 14 tribal nations. That could mean anywhere between 300,000 and 500,000 people, according to DOH.
Both the Pfizer and NIH-Moderna vaccine are given in two doses three and four weeks apart, respectively, and are reportedly 90% or more effective seven days after the second dose. Clinical trials revealed no major adverse effects, according to the U.S. Centers for Disease Control and Prevention.
At press time, six people in the U.S. are known to have had severe allergic reactions requiring treatment out of hundreds of thousands of shots administered.
The state plan for vaccine distribution follows the National Academy of Medicine’s Framework for Equitable Allocation of COVID-19 Vaccine. Teachers, child care workers, and other essential workers are next in line. “We’ll have most people in Washington vaccinated by mid-summer,” said Michele Roberts, the state’s acting assistant secretary of health.
“If 75% to 80% of Americans are vaccinated, then by the end of 2021, we can reach a degree of normality,” said Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases, referring to the threshold needed for achieving herd immunity without endangering anyone by further exposure to the virus.
For Washington state, that translates to over 10 million doses for more than 5 million people. Lower participation would mean it would take longer to stop the pandemic.
“We’re supposed to get it soon for staff and I will be administering it and definitely taking it,” said Anne Nesbit, KP fire department public information officer. “People have concerns and so do I … but someone has to lead. And we’re not dropping our guard; we have to keep up with masks and precautions to stop the spread.”
Dr. William Roes of Key Medical Clinic in Key Center said he has applied to become a vaccine distributor under the auspices of Community Health Care, which will be the clinic’s new owner in January.
“I don’t know if we’ll get it or not, but we’re in a unique situation (on the Key Peninsula), so maybe. It will be months though, after the first responders and so on.”
Roes said he understood suspicion about the speed of the vaccine’s development might make people wary about getting it. “What seems like a miracle now is really a product of how we dealt with SARS, MERS, with Ebola; the techniques and technology since then have transformed everything. I will take it as soon as I can get it,” he said.
A Gallup poll published Dec. 8 reported that 63% of Americans were willing to get the vaccine, up from 50% in September. When asked about the pace of the vaccine approval process, 78% said their primary concern “is that it will move too fast, without fully establishing safety and effectiveness.”
Traditional vaccines can take about 10 years to develop. The fastest vaccine — for mumps — took four years. Because of the severity of the COVID-19 pandemic, research efforts were boosted and clinical trials ran concurrently instead of consecutively.
A significant difference about both the Pfizer and NIH-Moderna products is that they are messenger RNA vaccines that do not include dead or weakened virus to trigger an immune response, and therefore don’t take nearly as long to produce. The mRNA contains a genetic code that mimics the virus to provoke the body into creating antibodies. The technology has been studied for more than a decade and does not affect a person’s DNA, according to DOH.
The success of any vaccine stopping the coronavirus pandemic depends both on people taking it and also continuing to wear masks and practice social distancing for many more months, according to Dr. Colleen Kraft, associate chief medical officer at Emory University Hospital in Atlanta, in a Nov. 25 interview with Today.
“I would say we have at least another year at the rate we’re going,” she said. “The more people that are just refusing to wear masks and making this an issue of personal freedom, I think the longer we’re going to be wearing masks. The longer we can’t get it done, the longer the pandemic rages on.”
The CDC and World Health Organization recommend wearing a mask even if individuals don’t feel sick because asymptomatic and pre-symptomatic people can spread the virus to others. It’s also unknown whether or not people who have received the vaccine can still transmit the virus.
A review conducted in June by researchers at the Oregon Health & Science University found no evidence of serious harm from wearing a mask.
“Masks can have some occasional adverse effects like discomfort, skin irritation or shortness of breath, especially for people with chronic lung or heart disease,” said Gopal Allada, M.D., associate professor of medicine at OHSU (pulmonary and critical care medicine). “However, on balance, the benefits in preventing virus transmission far outweigh the potential risks of wearing a mask.”
The report concluded that “even a small effect can have a big impact on reducing exponential spread of novel coronavirus.”
The U.S. sustained 323,000 deaths from COVID-19 by the middle of December. The U.W. Institute for Health Metrics and Evaluation estimated a total of 562,000 by the end of March. It expects that vaccines will have saved 25,000 lives by then — but that broader mask usage in this period could save 56,000.
Update: The Department of Health is determining what groups of people will have access to the vaccine first. More information is at https://www.doh.wa.gov, including their phased approach to vaccine allocation. Take the DOH survey to determine eligibility and sign up for notification at https://wadoh.jotform.com.
UNDERWRITTEN BY THE FUND FOR NONPROFIT NEWS (NEWSMATCH) AT THE MIAMI FOUNDATION, THE ANGEL GUILD, ADVERTISERS, DONORS AND PEOPLE WHO SUPPORT INDEPENDENT, NONPROFIT LOCAL NEWS